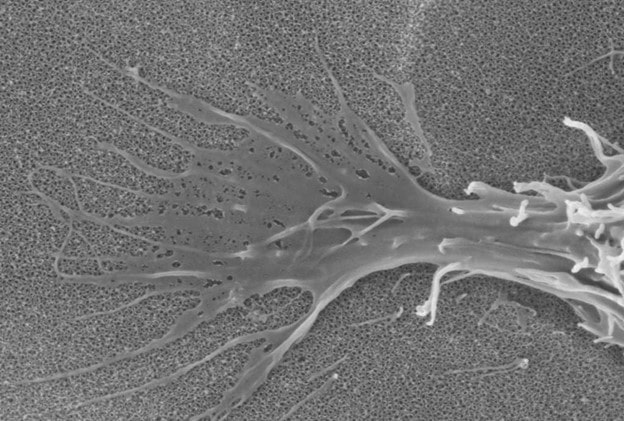
Intellectual Property Protection
Nanovis® maintains a robust intellectual property portfolio consisting of several owned or licensed patents. All nanoVIS Ti™ Surface Technology components are protected by a series of patents. Additionally, the proprietary process used to produce our unique surface is safeguarded by multiple trade secrets.
Regulatory Strategy and Pathway
Nanovis® has achieved several significant regulatory milestones:
NanoVIS Ti™ Surface Technology is the only widely available platform nanotechnology to achieve FDA's nanotechnology designation.
We are the first company worldwide to obtain FDA clearance and nanotechnology designation for a pedicle screw system.
We are the second company worldwide to receive FDA clearance and nanotechnology designation for an interbody fusion cage.
The Master File for nanoVIS Ti™ Surface Technology has been referenced for multiple 510(k) clearances across two different product types in spine.
Clinical and Commercial Advantages
To validate the nanoVIS Ti™ Surface Technology, Nanovis® has commercialized a portfolio of spinal products. Learn more at nanovisspine.com.
Clinical Advantages
- FDA nanotechnology designation. First company in the world with designation on PEEK + Ti interbodies (CP Ti) and Open/MIS pedicle screws (ELI Ti).
- Statistically significant and superior comparative surface data in the label – hMSC mineralization and osteoblast mineralization at 21 days, in vitro.
Commercial Benefits
- On-label marketing for superior positioning and messaging.
- Superior FDA label used to drive new surgeon conversions, hospital approvals, and technology price premiums.
- Surface-on-a-surface story — add a nano surface to a macro-porous 3D-printed surface or a micron-roughened base.
- Surgeon engagement at a scientific and innovation level, enabling studies, papers, podium presentations, etc.

Let’s talk nano surface
Join Our Newsletter
We’ll send you a newsletter once a month, no spam.